See Water and Heavy Water for thermodynamic properties at standard condtions. The density of water depends on temperature and pressure as shown below: Note! Temperature must be within the ranges 0-370 ☌, 32-700 ☏, 273-645 K and 492-1160 °R to get valid values. The output density is given as g/cm 3, kg/m 3, lb/ft 3, lb/gal(US liq) and sl/ft 3. The calculator below can be used to calculate the liquid water density at given temperatures. See more about the difference between mass and weight Online Water density Calculator The density of water is 1.940 sl/ft 3 at 39 ☏ (4 ☌), and the specific weight in Imperial units is 
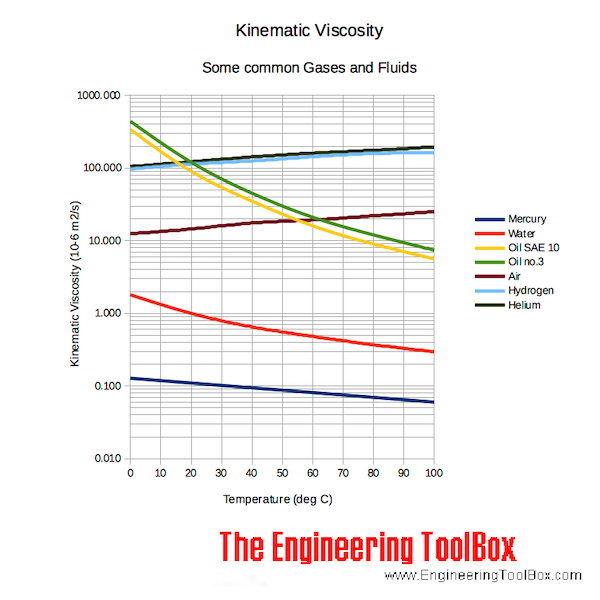
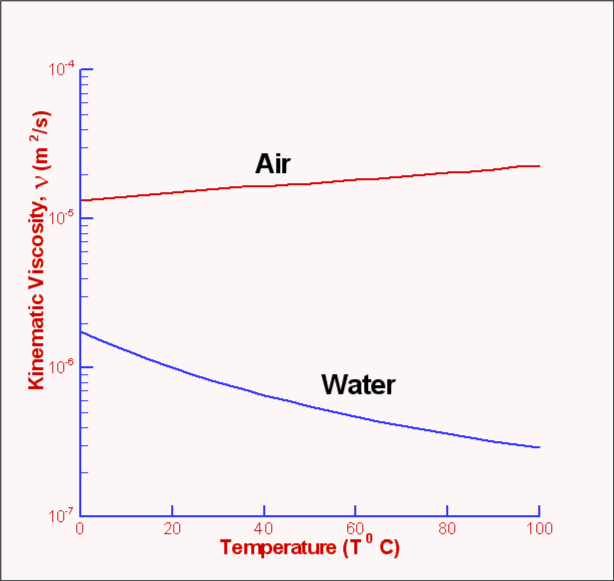
In the Imperial system the mass unit is the slug, and is derived from the pound-force by defining it as the mass that will accelerate at 1 foot per square second when a 1 pound-force acts upon it :ġ = 1 * 1 and 1 = 1 /1 In the SI system, specific weight of water at 4☌ will be: G = acceleration due to gravity, units typically and value on Earth usually given as 9.80665 m/s 2 or 32.17405 ft/s 2 Specific weight is the ratio of the weight to the volume of a substance: Pure water has its highest density 1000 kg/m 3 or 1.940 slug/ft 3 at temperature 4☌ (=39.2☏). Water - Properties at Gas-Liquid Equilibrium Conditionsįigures and tables showing how the properties of water changes along the boiling/condensation curve (vapor pressure, density, viscosity, thermal conductivity, specific heat, Prandtl number, thermal diffusivity, entropy and enthalpy).Density is the ratio of the mass to the volume of a substance: Temperature and Pressureįigures and tables showing thermal diffusivity of liquid and gaseous propane at varying temperarure and pressure, SI and Imperial units. Temperature and Pressureįigures and tables with Prandtl Number of liquid and gaseous propane at varying temperarure and pressure, SI and Imperial units. Temperature and Pressureįigures and tables showing Prandtl number of nitrogen at varying temperarure and pressure, SI and Imperial units. Hot and cold water service systems - design properties, capacities, sizing and more. Thermodynamics of steam and condensate systems. Steam & condensate systems- properties, capacities, pipe sizing, systems configuration and more. Material properties of gases, fluids and solids - densities, specific heats, viscosities and more. Involving velocity, pressure, density and temperature as functions of space and time. Prandtl number of water at given temperatures and 1, 10 and 100 bara (14.5, 1 psia): Water - Prandtl Number vs. Temperature at Atmospheric Pressure State Prandtl number of water at atmospheric pressure, temperature given as K, ☌ or ☏: Water - Prandtl Number vs. Prandtl number of water at 1, 10 and 100 bara (14.5, 1 psia), varying temperature given as ☌ or ☏: Prandtl number of water at 1 bara pressure, varying temperature given as ☌ or ☏: See also other properties of Water at varying temperature and pressure : Boiling points at high pressure, Boiling points at vacuum pressure, Density and specific weight, Dynamic and kinematic viscosity, Enthalpy and entropy, Heat of vaporization, Ionization Constant, pK w, of normal and heavy water, Melting points at high pressure, Properties at Gas-Liquid Equilibrium Conditions, Saturation pressure, Specific gravity, Specific heat (heat capacity), Specific volume, Thermal conductivity, Thermal diffusivity and Vapour pressure at gas-liquid equilibrium, and Thermophysical properties at standard conditions ,Īs well as Prandtl number of Air, Ammonia, Carbon dioxide, Methane, Nitrogen and Propane. K = thermal conductivity, īelow, Prandtl numbers of water at varying temperatures and 1, 10 and 100 bara (14.5, 1 psia) are given in figures and tables. Μ = absolute or dynamic viscosity, Ĭ p = specific heat, The Prandtl number can for calculations be expressed as The Prandtl Number - Pr - is a dimensionless number approximating the ratio of momentum diffusivity (kinematic viscosity) to thermal diffusivity - and is often used in heat transfer and free and forced convection calculations. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
